In recent decades, healthcare professionals and oncology specialists have become increasingly focused on the role of second hand smoke as a cause or contributing factor to the incidence of lung cancer in non-smokers. Studies which indicate a correlation between passive smoking and an increased risk for developing lung cancer can be used as evidence to encourage further public education and anti-smoking campaigns. Additionally, relatives and friends of smokers will have access to scientifically tested information, enabling them to make informed choices about their exposure to passive smoking. The purpose of the study is to evaluate whether there is a correlation between passive smoking and lung cancer.
In 2011, 226 lung cancer patients from Bangkok Lung Center were evaluated based on their occupation, exposure to passive smoking, family history of cancer and other co-morbidities. The studied sample consisted of men and women with late stage lung cancer who were admitted to Bangkok Hospital between January 1 - December 31, 2011. These patients had previously undergone chemotherapy and radiation therapy. The diagnosis was proven by lung pathology, and the patients were evaluated retrospectively. Data was collated using IBM SPSS statistics analysis. Researchers measured the collated data for indications of a significant amount of lung cancer occurrences within the group of females who had been exposed to passive smoking. The rate of 5% was chosen to indicate a significant occurrence.
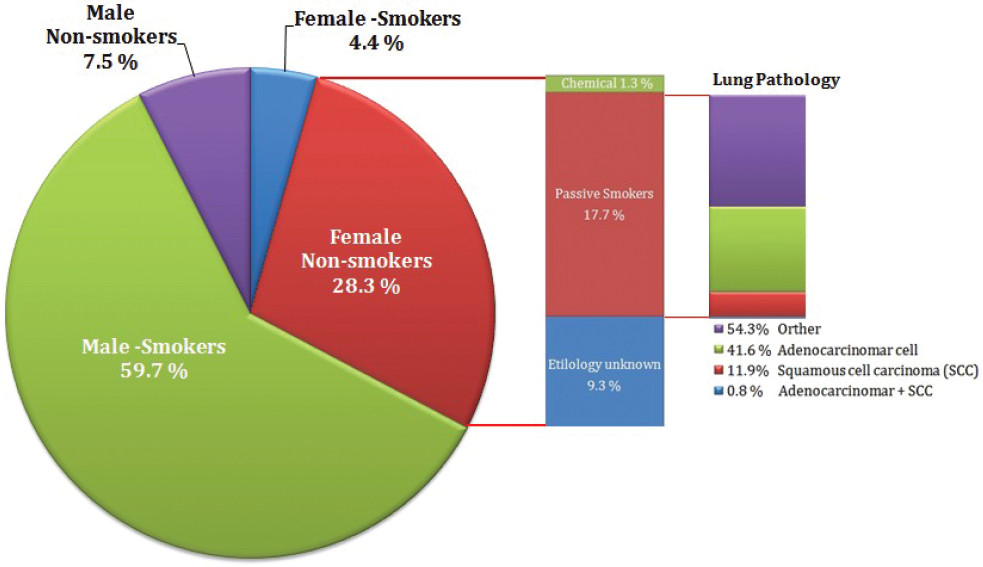
Figure 1: Statistics of male and female participants in the study
Table 1: Characteristics and Initial measurements of analysis subjects
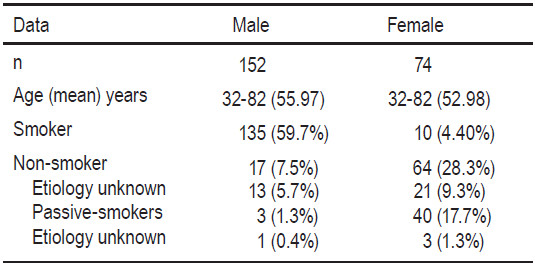
The study yielded the following results: of 152 male patients, 135 were smokers (59.7%) and 17 (7.5%) were non-smokers. The age ranged from 32-82 years. The mean age for male patients was 55.97 and the mean age for female patients was 52.98. The mean age for all patients was 54.99. All of the participating patients in the study were Thais. Among the 17 non-smoking males, 3 were passive smokers and 1 worked in a chemical factory. There were 74 females, 10 (4.4%) were smokers and 64 (28.3%) were non-smokers. Among 64 non-smoking females, 40 were passive smokers (p < 0.001) and 3 worked in a chemical factory (Figure 1, Table 1).
The occurrence of lung cancer in female passive smokers was found to be significantly high at 17.7%. Moreover, lung pathology revealed adenocarcinoma as the most common type of cancer in this group (41.6%). Cases of squamous cell carcinoma (SCC) amounted to 11.9% and 0.88% had both adenocarcinoma and SCC.
Lung cancer is diagnosed more frequently than any other type of cancer in both men and women, and while smoking remains the predominant cause, recent studies reveal that lung cancer rates among people who have never smoked are higher in women than in men.1 In industrialized countries, lung cancer rates have a direct relationship to smoking behaviors. In Asia, particularly in China, the pattern varies such that lung cancer rates in men reflect high smoking rates but high rates among non-smoking women appear to be related to other factors.2 The following were identified as potential risk factors: cooking oil vapour, exposure to environmental tobacco smoke (ETS), occupational hazards such as exposure to toxins and chemicals i.e. asbestos and ambient air pollutants.
Northern Thai women have one of the highest incidences of lung cancer in Asia. The incidence rate, however, differs significantly from region to region despite similar low smoking prevalence, lifestyle factors and diet. Several studies were conducted, including: a study of local sources of drinking water,a mutagenicity test of urine samples, and fungi and bacteria were measured in indoor living areas. The fungus Microsporum canis was identified as a possible cause of the high incidence of chronic respiratory diseases among women in Sarapee. The women had an elevated concentration of the fungus serum antigen, which was commonly found in Sarapee indoor air.3
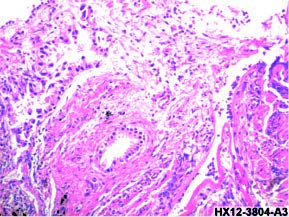
Figure 1: Microscopy: Infi ltrating groups of malignant cuboidal to columnar epithelial cells with glandular formation are seen in chronically infl amed anthracotic bronchial and lung tissue biopsies. Moderate nuclear pleomorphism of the malignant cells is noted.
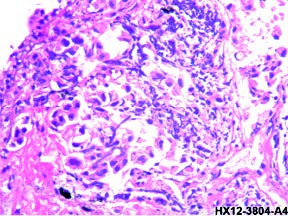
Figure 2: Immunohistochemistry is positive for CK7, CEA, TTF-1,lung adenocarcinoma is confi rmed on both histopathology and immunohistochemistry.
In 2011, a study in Morocco4 yielded slightly similar results. The studied sample consisted of women with lung cancer who were admitted between January 2004 and December 2008. The diagnosis was proved by anatomo- pathologic analysis of biopsy specimen. The study found 101 women among 1,680 cases of lung cancer. Percentage of never-smokers was 75%, 14% were passive smokers and 11% are smokers. The proportion of non-small cell lung cancer (NSCLC) at stage IV was higher (82%); 16% of cases were at stage III and 2% were at stage II. The proportion of adenocarcinoma (ADK) among never and passive smokers is higher than squamous cell carcinoma (SCC) (69.4% vs. 30.6%), while among women who are smokers, the most frequent histological type is SCC (63.6%). ADK appears to be the most frequent cell type among never and passive smokers. ADK is significantly associated with lower rates of survival.
Lung adenocarcinoma is the most common primary carcinoma arising in women. For reasons not examined within the scope of this particular study, but potentially due to changes in smoking habits, adenocarcinoma has replaced squamous cell carcinoma as the most common primary lung carcinoma in present days (Figures 2 and 3).
The study confirms the significant correlation between passive smoking and lung cancer. In 2007, 11 million of the 51 million people in Thailand aged over 15 years old were identified as smokers.5 This delineates a 1:5 ratio of smokers to non-smokers. Families with smoking family members have the greatest risk of exposure to second-hand smoke. Wives who have smoking husbands have a higher risk of developing lung cancer than women who have non-smoking husbands.6 Other risks include occupational hazards such as exposure to toxins and chemicals, i.e. asbestos and ambient air pollutants.
Since the health risks from asbestos exposure increases with heavier exposure and longer exposure time, patients who have worked around carcinogen-exposed workplaces are at higher risk. However, researchers have also found asbestos-related diseases in individuals who only experienced brief exposures. Generally, those who develop asbestos-related diseases show no signs of illness for a long time after their first exposure.7
A study published in 2008 conducted a cross-sectional exposure survey among nonsmoking women and children in Eastern Europe, Latin America, Asia and the Middle East. The research used passive air monitors to measure household air nicotine concentrations and a hair nicotine test to measure personal exposure to passive smoking.8 After surveying a total of 1,284 households in 31 countries including Laos, Cambodia, Thailand and China, findings suggested that the number of smokers in a household is directly related to the level of hair nicotine concentration. When there were a larger number of smokers, the level of nicotine concentration in the hair was higher, thus creating an elevated risk for developing tobacco smoke exposure-related diseases. A similar study was done in urban and rural China, measuring nicotine levels specifically in public places. Airborne nicotine was detected in 91% of the locations sampled and provided evidence for the need to protect the public from exposure to passive smoking.9
In India, there have only been a few studies on the pulmonary effects of passive smoking. One study examined 9,090 adolescent school pupils and discovered that exposure to environmental tobacco smoke (ETS) was correlated with an increased risk of asthma. Another study showed that ETS exposure was a trigger for an acute exacerbation of asthma. Passive smoking subtly affects airflow mechanics. Another study found a correlation between passive smoking and lung cancer. In this study,50 healthy non-smoking women exposed to passive tobacco smoke were matched to 50 women of the same age who had not been exposed to tobacco smoke. In a study carried out in collaboration with the International Agency for Research on Cancer, exposure to ETS during childhood was strongly linked to an higher rate of lung cancer (OR = 3.9, 95% CI 1.9-8.2). In conclusion, passive smoking can lead to several adverse pulmonary conditions, just as seen in other countries; this is also reported in India.10
The results of this study identify a significant risk factor for developing lung cancer: exposure to passive smoking. These results also support the conclusions of other studies conducted internationally. The evidence is clear: continued exposure to passive smoking signifi- cantly increases the risk of cancer. The continuance and strengthening of public health campaigns to discourage smoking is necessary to combat this growing public health risk. The population needs to be further educated about the dangerous risks associated with passive smoking.
Furthermore, a re-evaluation of lung cancer screening guidelines is recommended. The current National Comprehensive Cancer Network® (NCCN®) Clinical Practice Guidelines in Oncology (NCCN Guidelines®) have been updated in 2012 to include recommendations for lung cancer screening from a new Lung Cancer Screening Panel.11 While their recommendations are up to date, especially in the use of technologically-advanced equipment for the detection of cancer in the earliest, most curable stage, screening should be expanded to include additional risk groups (such as passive smoke). In light of the increase in the occurrence of lung cancer in females exposed to passive smoke, more comprehensive screening guidelines would assist medical professionals in increasing the detection of developing cancers. Additionally, based on our finding the guidelines’ recommended age to begin routine screening should be performed in earlier age group (< 50 years old) particularly for those with passive smokers.
The NCCN Guidelines provide:
−Individuals without symptoms of lung cancer be assessed for risk based on the following factors: age, smoking history, radon exposure, occupational exposure, cancer history, family history of lung cancer, lung disease history, and secondhand smoke exposure.
−Screening with helical LDCT is recommended for patients at high risk, defined as:
−Age 55 to 74, with 30 or more pack-year history of smoking tobacco (category 1); if a former smoker, must have quit within 15 years (category 1)
−Age 50 or older and 20 or more pack-year history of smoking and one additional risk factor (other than secondhand smoke) (category 2B)
−Routine lung cancer screening is not recommended for the moderate-risk group, defined as age 50 or older and 20 or more pack-year history of smoking or secondhand exposure and no additional risk factors, or for low-risk individuals, who are under 50 and/or have a less than 20 pack-year history of smoking.11
The data in this study indicates that the mean age for all patients was 54.99. Considering that these are advanced stage cancer patients, the development of the disease is presumed to have begun approximately 10 years before this evaluation. Adhering closely to the current guidelines could potentially increase the number of undiagnosed cases.