Lipid lowering agent in statin group or 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors has been used for reducing the lipid level in the blood, and is useful for preventing patients from coronary artery diseases (CADs). Statin are able to decrease the disability rate and death rate among CAD patients. It can reduce the low-density lipoprotein cholesterol (LDL-C) effectively and pleotropic effects which cause the forming of plaque stabilization. In addition, statin drugs can decrease the triglyceride (TG) rate in the blood stream by about 30% to 50%, and it can increase the high-density lipoprotein cholesterol (HDL-C) rate in the blood stream by about 5% to 10%.1 Although statin is considered as effective and safe, there are some patients who stop using this drug because of the undesirable effect on the muscles which is found often in statin users. From observational studies,1,2 the reports show that the undesirable effect rate from using statin drugs on muscles was at 10% to 15% of total treated patients. But in 2014, there were the meta-analysis studies which collected moderate undesirable effects to muscle incidents, for example, the muscle pain symptom, which is not related to rhabdomyolysis, may not find the abnormal rate of CK, which is called myalgia, or if the symptom is related to the higher rate of CK at least 10 times of normal upper value, which is called myopathy. In these studies, the undesirable effect rate in the statin sample group was the same as the placebo group.3 If the patient continues to use statin drugs, the condition may develop to rhabdomyolysis. The symptoms include severe muscle pain, muscle necrosis or myoglobinuria associated with CK level increasing to 40 times. The report of this incidence has been found in about 1-3/10,000 treated persons-year.4 However, this undesirable effect is the main reason for a patient to discontinue using statin drugs or to affect the cooperation of a patient to use statin.5 Most of the patients have less symptoms or are back to normal conditions after stopping using statins. Nevertheless, there are some patients whose symptoms still exist and are getting more severe. It could be assumed that the patient has developed autoimmune myopathy. The biopsy procedure could be performed to investigate muscle necrosis and autoantibodies against HMG-CoA reductase, which requires treatment by immunosuppressant drugs.6,7 This article aims to review the epidemiology, the mechanism of action, and the different statin-associated muscle symptoms.
There are many undesirable effects of statin-associated muscle symptoms, even though the certain definition of this condition has not been defined yet.8 Most patients have muscle complaints (muscle pain, aching, myalgia, weakness, stiffness, tenderness, or cramping related to nocturnal cramping) together with flu-like symptoms and a normal CK level. These symptoms are called myalgia. In some patients, we might find the symptom called myositis, which is related mainly to myalgia, myonecrosis, and high levels of CK over the normal upper limit. There are 3 stages of SAM:
1) Mild level: CK level is at 3-10 times higher than the upper normal limit;
2) Moderate level: CK level is at 10-50 times higher than the upper normal limit (the symptom which is associated with a CK level higher than the upper normal limit over 10 times is called myopathy);
3) Severe level: CK level is at 40-50 times higher than the upper normal limit.
Other symptoms could be found associated with the previously mentioned symptoms such as weight loss without any indications, tachycardia, nausea, and red-brown color to urine due to myoglobin dissolving. In some patients, symptoms could develop to be more severe, leading to rhabdomyolysis. The patient would suffer from severe muscle pain, muscle necrosis, or myoglobinuria associated with higher creatinine serum and a CK level over 40-50 times than the upper normal limit. These aforementioned symptoms are called in general Statin-associated Muscle Symptom (SAM).8-10 Most of SAM patients have symmetry muscle pains or weaknesses, which is often found in hips, thighs, calves, and back muscle. The incidents of the side effects are different in each patient. The side effects could be found in the first 4-6 weeks after using statin drugs, or found later after years of using statin drugs and such symptoms also could be found in patients who take a higher dose or that interact with other drugs.11
SAM has been considered as a problem in medicine since 2001. Cerivastatin has been withdrawn from the market because of reports of rhabdomyolysis and the death of cerivastatin users. However, at present, there is still no full definition of SAM. The incidents from clinical trials are very low (1.5%-5%). The report of rhabdomyolysis has shown about 0.01%-0.04% of total cases. From the 42 randomized and controlled trials it was found that there was no difference in SAM risk, from statin users and placebo-statin users. In the meantime, an observation study found that SAM was the main reason for discontinuation of statin drugs in statin users.3 Interestingly, there were higher numbers of reports of SAM in general practice than in clinical practice. This might be due to the selection process of clinical practice in selecting SAM-associated symptom patients and the interaction of statins with other drugs. These patients might be disregarded from the study group which could explain why there were no reports of SAM in those studies.12 In addition, there are patients own factors and amount of dosage uses which affect SAM. SAM is likely to be found in high dosage statin users.13 Today, the Food and Drug Administration of USA (FDA) has warned against the use of 80 mg simvastatin in all patients.13,14 In Thailand, between 1996 and 2009, there were 198 report cases of SAM, most of these patients were diagnosed as rhabdomyolysis (55.6%). However, mild SAM may not be reported in these studies. The studies shown related factors to SAM which included ageing patients, female gender, associated diseases, and the interaction of statin with other drugs.15
There is little and very limited knowledge for the mechanism action of SAM. However, it has become clearer more recently. The assumptions for the mechanism of SAM are:
- The decreasing of isoprenoid; isoprenoid is the fat compound which is produced from the HMG-CoA reductase pathway. It mainly consists of farnesyl pyrophosphate (F-PP) and geranylgeranyl pyrophosphate (GG-PP). When F-PP and GG-PP decrease, it causes the membrane protein (prenylated proteins), such as GTPase to become unable to attach to the cell membrane, which causes the abnormal reception in cells. These will increase cystolic calcium which stimulates capsase-3 and sends a signal to cells to cause apoptosis or abnormal gene expression.16 Furthermore, the decreasing of isoprenoid can cause the reduction of coenzyme Q10, which is an important substance for the mitochondrial electron transport chain. Therefore, using statin could have the side effect of catalyzing muscle energy. However, no evidences has as yet given proof of the relationship between the decreasing of coenzyme Q10 and muscle disorder.13 There were only a few studies reporting the benefits of taking coenzyme Q10 100 mg per day, which could reduce the severity of SAM patient in daily life, but did not decrease CK level.17,18
Nevertheless, the recommendation for treating patients with coenzyme Q10 in SAM patient is still being debated, as the coenzyme Q10 deficiency can be found in older age patients, and high dose statin patients or patients who have high levels in statin bioavailability from a liver or kidney disorder, or metabolic syndrome patients such as familial mitochondrial encephalomyopathy. Treating this group of patients with coenzyme Q10 might decrease SAM. However, there is no evidence found recently to prove the use of coenzyme Q10 can prevent SAM.19
- The decreasing of cholesterol in sarcolemal and sarcoplasmic reticular affects the imbalance of membrane and plasma lipids. Thus, the membrane lipids are unstable. However, there are some studies debating this assumption which looked into the inhibition of squalene synthetase in the myotubule of muscle cells.20
- The disturbed intracellular calcium homeostasis; calcium has an important role in controlling the function of muscle cells. L-type calcium channel is the medium channel for increasing calcium levels in cells. Ryanodine receptors which are located in the sarcoplasmic reticulum send the signal to create action potential and muscle contraction. The study of muscle cell biopsy in statin users and muscle weakness patients found that ryanodine receptors 3 (RR3) were increased.8
- The dissolution of muscle cells, stimulated by statin drugs, has been linked to the human leukocyte antigen (HLA) allele class II DRB 1*11:01 related to the production of anti-HMG-CoA reductase autoantibodies. HLA DRB1*11:01. Patients are likely to develop a muscle disorder 25 times more in white patients and 50 times more in black patients than other patients who do not have this kind of antigen. In addition, after the muscle cell is exposed to statin drugs, it will abnormally increase the results of HMG-CoA reductase and the number of HMG-CoA reductase protein. This may cause an autoimmunity condition and the interaction between statin drug and HMG-CoA reductase can cause protein formation which is considered as a foreign substance.21 Furthermore, the study of muscle biopsy in severe myopathy patients or persistent muscle disorder patients after the discontinuation of statin use found that there is muscle necrosis and the presence of autoantibodies against HMG-CoA reductase.21
The diagnosis of SAM should consider surrounding factors as well. It may be cause mainly by other factors which are not related to SAM, for example, a mistake from lab testing (abnormal CK), a previous condition of patient, a concomitant drug, a conformity of symptoms and taking period, and symptoms presenting after de-challenging or re-challenging statin drugs. Nowadays, there is no gold standard diagnosis for SAM.11
The healthcare professional team has to consider other possible factors that might cause the symptoms. They have to review the indications of using statin,11 then set a timeline for patient evaluation and communication between the healthcare professional team members. There are many possible factors that cause these symptoms, however, from the relative risk study of each of the factors, studies have not yet confirmed the real cause.9,22 The healthcare professional team should also consider other risk factors in the diagnosis of SAM. The risk factors for SAM are mainly considered by the following:
I. Patient factors
1. General conditions; aging, female gender, or low body mass index (BMI) patients have a greater risk of myopathy.9 This includes some activities which cause muscle disorder such as extensive exercise or abnormal position of muscle.11
2. Patients with a medical history of muscle disorder or joint pain before taking statin, including repeated cramp, using other lipid lowering drugs, or having a family member with a medical history of myopathy which is related to lipid lowering drugs. In patients who are diagnosed with amyotrophic lateral sclerosis (ALS),9,23 after taking statin, an increasing number of cramps and SAM were reported. Patients who have polymyalgia rheumatica are also in this category.11
3. Other underlying conditions in patients, such as hypothyroidism which causes secondary dyslipidemia is an important risk factor that causes SAM. If the patient has SAM, the investigation for thyroid function should be performed.8,11 Chronic renal failure patients are at a greater risk for severe myopathy called rhabdomyolysis.24 For hypothyroid patients who have reported SAM, the symptoms can disappear after statin discontinuation, but in some cases, the patient may need to stop taking statin and be given a thyroid hormone until the thyroid can perform functionally.24 Medicines or drugs which affect the muscles such as corticosteroids, psychiatric drugs (risperidone or haloperidone), immunosuppressive agents, alcohol, or cocaine might cause symptoms.
4. The genetic marker for the statin receptor or enzyme cytochrome P (CYP) should be considered; all statins except fluvastatin transport into the liver cells through an organic anion transporting polypeptide or OATP1B1 which is controlled by the SCLO1B1 gene. According to the data from SEARCH (Study of Effectiveness of Additional Reductions in Cholesterol and Homocysteine collaborative group) it was shown that there is a relationship between SAM ratio and the statin genetic differentiation in the liver cell. It was found that myopathy incidence correlated with non-coding single nucleotide polymorphisms (SNPs) at intron number 11 on SCLO1B1 gene or rs4363657 location. When replacing allele with C at 1 position, the possibility that a myopathy incident would occur was up to 4.3 times (95% CI = 2.6-7.2). However, if the allele is replaced with C at 2 positions (CC homozygous), the possibility of a myopathy incident occurring rises up to 17.4 times (95% CI = 4.8-62.9) comparing to TT homozygous.8,25 Thereafter, from the STRENGTH study (Statin Response Examined by Genetic Haplotype Markers), data collected showed the changing of CYP2D6, CYP2C8, CYP2C9, CYP3A4 and SLCO1B1 genes which the finding
that SLCO1B1*5 has a clear correlation with SAM. Myopathy incidents can occur, when the allele is fixed, in up to 17% of cases. In the meantime, if replacing 1 position and 2 position, SAM can occur up to 27% and 50% respectively (p=0.01).8,26 CYP enzyme has an important role in the first step in the metabolism of statin drugs. Nowadays, we find that there each CYP enzyme has more than 30 isozymes. Each statin has a different CYP isozyme. The differentiation in the genetic CYP enzyme has a certain effect in drug metabolism. There are 2 factors, which affect drug metabolism:
1) Patient takes the drug which then metabolises through the CYP enzyme in the same way as a statin. It changes the function of the enzyme to match with a metabolism result which is different from the normal process.
2) The statin which metabolises through the glucuronidation process, and does not relate to CYP enzyme function.8 However, genetic testing is very costly and has to be performed in a specialty laboratory; thus, testing for the differentiation of genetic receptors and enzymes is not popular in general medical practice.
II. Drugs factor
Each statin has different characteristics; please see Table 1. Hydrophilic or lipophilic properties are the factors for muscle disorders. Pravastatin and rosuvastatin are hydrophilic drugs and need OATP to transport it in to the liver cell, which dissolves into myocyte insignificantly.8 From all reports of muscle disorder incidents, hydrophilic-statin drugs are reported in very few cases. According to data from 19,592 patients, no difference was found between the pravastatin group and the placebo group in the level of creatinine phosphokinase (CPK). There were no reports for myopathy or rhabdomyolysis in this group.27 However, for lipophilic statins, they can enter the cell freely, therefore, they are likely to enter the liver cell, myocyte cell and other cells quickly. This action can cause muscle disorders for patient in this group.
The characteristic of hydrophilic and lipophilic statin
Table 1: Outline of the primary search findings.
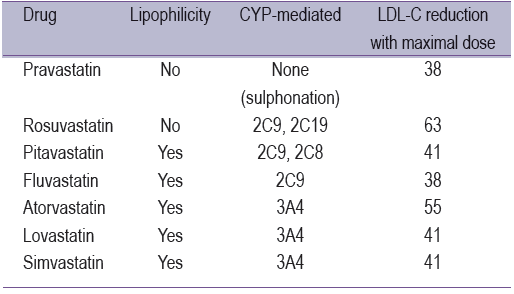
drugs is shown in Figure 1,28 which found that cerivastatin is the best for lipophilicity. This drug has been withdrawn from the market because of the high numbers of incident reports for rhabdomyolysis. Many reports regarding myopathy have been due to high doses of simvastatin (80 mg). Therefore, the high dose of statin drugs might be another factor to consider. In addition, the interaction between stain and other drugs might cause muscle disorder incidents too.29
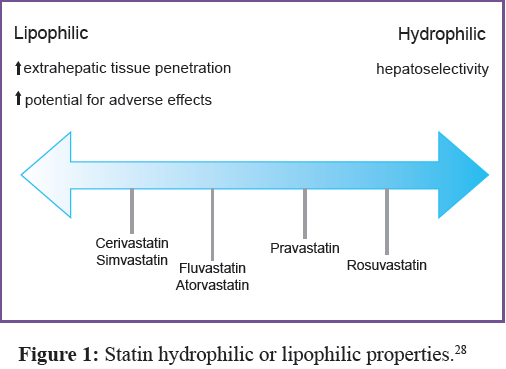
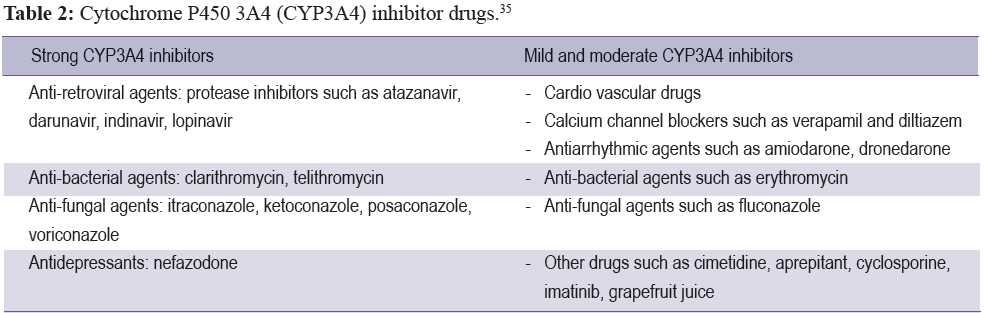
For patients who have to take many medicines at the same time, they are more at risk for myopathy than others, as some drugs might inhibit the CYP450 enzyme to be active, especially CYP3A4 which causes the level of statin drugs to increase. Each statin drug has a different kind of CYP metabolism. Simvastatin, lovastatin, and atorvastatin are metabolized by CYP3A4. Therefore, if the patient takes strong CYP3A4 inhibitors it is likely to increase the incident rate of rhabdomyolysis symptoms. With a mild to moderate CYP3A4 inhibitor (Table 2), it is still possible to find myotoxicities, while some part of fluvastatin is metabolized by CYP2C9. In addition, the concomitant interaction between statin and fibrate or niacin is likely to increase the incident rate of muscle disorders as well, and this includes colchicine drugs.30-32
The common evaluation of SAM is pain and weakness. Most patients have pain at symmetrical proximal muscles such as thighs, hips, calves, and back.33 The symptoms could be found in the early stage of taking statin or years later after using statin. Most of the patients have the symptom affecting the back after taking statin for 4-6 weeks and the symptom could exist for years. This symptom could be found after increasing statin dose or taking a concomitant drug which elevates the level of statin, or extensive exercise in a patient. If these patients discontinue using statins, the symptom may disappear. However, if the patients re-challenge the same statin, the muscle disorder could occur again immediately. Nevertheless, 90% of these patients could elect to switch to other statin drugs without leading to any muscle disorder symptom.11,34
The level of CK is a result from alaboratory test which reflects the operating function of muscle, heart muscle and brain. Testing the CK level before starting statin drugs is not necessary for all patients. The American Heart Association and National Heart, Lung and Blood Institute recommends checking CK levels before taking statin. However, the National Lipid Association’s muscle expert panel agrees that checking CK levels before taking statin is a lavish expense. According to this information, it is recommended for patients who are high risk only, who are: aged 75 years old and over, patients who take concomitant drugs, have a medical history of kidney and liver diseases. However, it is not recommended to follow up CK levels in every patient. It is recommended to focus only on the patient who has a record of muscle disorder to evaluate or diagnose SAM.11
However, higher CK levels may not be caused by statin drugs alone,36 it may be caused by other factors such as excessive exercise. It is recommended to monitor symptoms closely. The health professional team should be cautious to carefully diagnose SAM in this group of patients and follow up the CK level at the appropriate time.8,11 In summary, after the discontinuation of taking statin drugs or concomitant drugs within 2-3 weeks, the symptom of suspected SAM patients should show improvements and CK levels should reach the normal upper limit. The re-challenging of using statin drugs might cause the symptoms again within 1 month depending on the type or the dose of statin. If it is necessary to confirm SAM symptoms, it might be wise to test the patient with a placebo drug. However, it is not practical to run through that process with each patient.11
The patient, who still has abnormal CK levels and/or severe symptoms after a long period of discontinuation of statin, needs to be investigated further for other causes and be treated according to the finding of the results. However, if there are no other causes than taking statin, it may be a case of statin-associated autoimmune myopathy. It is recommended to test electromyography or perform the cell biopsy from the symptomatic muscle. For statin-associated autoimmune myopathy patients, findings could include muscle-cell necrosis, infiltration of macrophages and anti-HMG-CoA reductase autoantibody, which are not found in SAM patients. Presently,the false positive of enzyme-linked immunosorbent assays for anti-HMG-CoA reductase are to be found at around 0.7%.21
There are many guidelines for treating SAM patients. The healthcare professional team needs to balance the benefits for cardiovascular diseases and SAM severity. To benefit cardiovascular diseases treatment, statin instruction needs to be reviewed, and also the recommendation to change a patient’s lifestyle should be made.11
1. A myopathy patient who has a CK level more than 4 times the upper limit of normal (ULN), but less than 10 times of ULN, and if the patient has low risk of cardiovascular disease, it is recommended to discontinue using statin. However, if the patient has a high risk of cardiovascular disease, it is recommended to continue using statin and monitor CK level closely. If it is found that the CK level is higher than 10 times of ULN, it would be recommended to discontinue using statin until CK level is less than 10 times of ULN. Then, the patient should take a lower dose of statin and the patient again should be monitored closely. If the CK level is still high, the health professional team should consider investigating other factors, such as hypothyroidism, metabolic muscle disease or excessive exercise.
2. A myopathy patient who has a CK level higher than 10 times of ULN, if there are none of the other factors other than SAM and the patient has a high risk of cardiovascular disease, it is recommended to discontinue using statin because of the high risk of rhabdomyolysis. If the CK level reaches normal level, the patient can be switched to other statin drugs. It is also recommended to start at a low dose and to monitor CK levels closely. The patient, who still has the symptoms and laboratory results relating to rhabdomyolysis, is strongly recommended not to undertake re-challenging with statin.
After the discontinuation of statin and once the CK level is back to normal, if the statin re-challenge is considered, the patient can start with a low dose, or switch to a hydrophilic statin such as pravastatin or fluvastatin. The patient should start at a low dose, and increase slowly until the LDL-C reaches the target level without any signs of myopathy. If the myopathy occurs, it would be recommended to treat with intermittent dosing, which is taking the drug every other day or twice a week. From previous studies, the intermittent dosing treatment should be prescribed for high intensity statin which has a long half-life, such as atorvastatin, rosuvastatin or pitavastatin. From previous studies, it was found that the treatment can decrease the LDL-C level by about 12% - 38%, and 70% of the patients in this group has no reported myopathy.11,37,38
In the case that the patient cannot tolerate statin or the statin is not successful in decreasing LDL-C to target level there are several approaches. For the high risk cardiovascular disease patient, it is recommended to use ezetimibe as the first choice. However, if the patient still cannot tolerate this statin or the treatment is not successful after taking ezetimibe, it is recommended to use a single drug or combine with other drugs such as bile acid sequestrants or fenofibrate (not recommend for gemfibrozil). Niacin is not recommended to be used because there is no supportive evidence for the benefits to cardiovascular disease patients. In addition, undesirable side effects when using niacin have been reported. Nowadays, there are other medicines to choose from for the patients who do not succeed with conservative treatment. The medicines in this group are proprotien convertase subtilisin kexin type 9 (PCSK9) monoclonal anti body therapy,39 or products from red yeast rice (Monascus purpureus) – which has been reported successful for reducing LDL-C level. The red yeast product has a mechanism action similar to lovastatin, which reduces the cholesterol production by the liver. However, there is not much supported evidence of the benefits for the cardiovascular disease patient and investigation into any long-term side effects. Since the mechanism action of red yeast is the same as statin drugs, thus, it might cause muscle disorder too.11,40
Furthermore, there are some other options for managing SAM patients such as taking ubiquinone or coenzyme Q10 to compensate in muscle tissue. Nevertheless, from meta-analysis reports, treating with coenzyme Q10 does not decrease SAM symptoms or muscle disorders. There are only a few reports to support those claims clearly. Even treating with vitamin D is being debated. Patients who see the benefit of treatment with vitamin D mostly are vitamin D deficiency patients.11,41
After the discontinuation of statin drugs, for those who are diagnosed with statin-associated autoimmune myopathy, these patients need to be monitored closely for myopathy symptoms. If symptoms are more severe, the patient is recommended to take prednisolone 1 mg per 1 kg of bodyweight per day. If it is a mild symptom, it is suggested to prescribe together with other immune-suppressant drugs such as methotrexate, azathioprine, mycophenolate mofetil. If within 8-12 weeks of treatment, the symptom still exists and does not respond to treatment, it might be recommended to give intravenous immune globulin to the patient. After the condition of the patient has returned to normal, the health professional team should be aware of any recurrence of the symptom or other underlying diseases, and decrease the immune-suppressant drugs slowly.
Lipid lowering agents in the statin group or HMG-CoA reductase inhibitors are considered as important drugs for reducing the lipid level in the blood stream and have the benefits for preventing cardiovascular diseases. The main problem in the use of statin is myopathy or muscle disorder which has been reported in many cases, in about 10%-15% of all patients. Even though it may not be considered as severe side effects, this symptom could become a more severe condition called rhabdomyolysis. The symptoms could turn out to be more severe myopathy, muscle necrosis or myoglobinuria, including the incidence of the level of CK becoming higher than 40 times of ULN, which has been reported for 1-3/1,000 treated personsyear. The health professional team must take up the role to evaluate and diagnose the symptoms to determine whether it relates to statin use or not. Furthermore, the benefits and risks for taking statin should be considered together with the appropriate treatment plan for each patient.