Alife expectancy of the world population increases, there are more diseases that can occur in the elderly. Among those cataracts and glaucoma are categorized in the group of diseases related to longevity. Both cataract and glaucoma cause blindness in Thailand and worldwide.1-5 Nowadays, cataract surgery has developed into phacoemulsification because of the small surgical wound, rapid visual recovery and the suitability for glaucoma patients. Accordingly, it does not affect conjunctiva in surgery and filtering surgery occurs after phacoemulsification. Therefore, there are the studies which found that phacoemulsification affects a drop of intraocular pressure.6-10 However, there is a difference in the resulting decreasing IOP,11-13 and there is an argument for specific patients that can benefit from phacoemulsification.
In addition, there are limited studies on the long term change of IOP after cataract surgery in Thailand. The average patient follow up was less than 1.5 years.14-17 Thus, the researcher is interested in long term IOP change after phacoemulsification with intraocular lens implantation in glaucomatous andnon-glaucomatous patients.
A retrospective study of patients who have undergone uncomplicated, clear cornea phacoemulsification with intraocular lens implantation by the same surgeon (CS) at Panyananthaphikkhu Chonprathan Medical Center between February 2011 and February 2015. The study was approved by the Ethics Committee in human research from Panyananthaphikkhu Chonprathan Medical Center. There are 208 patients in the study, divided into 3 groups:
1. Nonglaucoma (NG)
2. Primary open-angle glaucoma (POAG) 3. Primary angle-closure glaucoma (PACG)
POAG was defined as eye with open-angle (posterior trabecular meshwork was visible for 360 degrees on gonioscopy), untreated IOP more than 21 mmHg and glaucomatous optic neuropathy with corresponding visual field loss. For PACG, those who have a close angle of eyes (appositional contact between the peripheral iris and trabecular meshwork over and equal 180 degrees), untreated IOP more than 21 mmHg and glaucomatous optic neuropathy with corresponding visual field loss. However, patients not included in the study include those with a postoperative follow up of less than 3 months, previous intraocular surgeries, and patients with secondary glaucoma.
The data including age, gender, and laterality of surgery, were recorded. If the patients had phacoemulsification in both eyes, the following data were recorded on the first surgery: preoperative and postoperative best-corrected visual acuity (BCVA) by Snellen chart, converted to a logarithm of theminimal angle of resolution unit (LogMAR) and; the number of antiglaucoma drugs. Goldman applanation tonometry was performed before surgery, 1 and 3 months postoperatively and then a final measurement of the study eye was recorded an average of 40 months after surgery.
Cataract surgeries were performed by one surgeon (CS) using the INFINITY vision system (Alcon Laboratories, Inc., Fort Worth, TX, USA). After topical anesthesia was
administered, a 3.0 mm temporal clear corneal incision, continuous capsulorhexis, hydrodissection, phacoemulsification, and aspiration of residual lens cortex were performed. A foldable IOL (AcrySof IQ SN 60 WF) was implanted in the capsular bag, and the viscoelastic was washed out. Postoperativetreatment consisted of 1% prednisolone acetate and 0.5% levofloxacin eye drops four times a day for 3 weeks.
Data were presented as mean ± standard deviation (SD) for continuous variables and number (%) for categorical variables. Repeated measure ANOVA and post hoc by Scheffe test was used to compare changes in intraocular pressure and vision. Paired t-test was conducted to determine the relationshipbetween preoperative IOP and postoperative IOP. A p-value of less than 0.05 was set as statistically significant.
According to the data collection from 208 participants, the average age of patients was 70.02 ± 8.96 years, 56% were women. Patients were divided into 3 groups as follows: no glaucoma (NG) 86 cases, POAG 61 cases, and PACG 61 cases. General baseline characteristics of patients in the three groups were comparable, except there were more woman in the PACG group. This matches epidemiology results of the previous study.2 (Table 1)
The mean baseline IOP in NG was lower than the glaucoma group (p < 0.001) and mean baseline IOP was similar in the glaucoma group (p =0.095). After phacoemulsification, there was a statistically significant reduction of the mean IOP at 1, 3 months and last follow up (mean 41 months). The postoperative mean IOP reduction was not different between eyes with open and closed-angle (p = 0.910 at one month, p = 0.229 at three months, and p = 0.081 for the last follow up) but a significantly greater reduction in postoperative IOP in the PACG group when compared with NG group at varioustime points (p = 0.049 at one month, p = 0.043 at three months, and p = 0.001 for the last follow up). Table 2 summarizes the change in IOP in all groups.
Table 1: Patient Demographics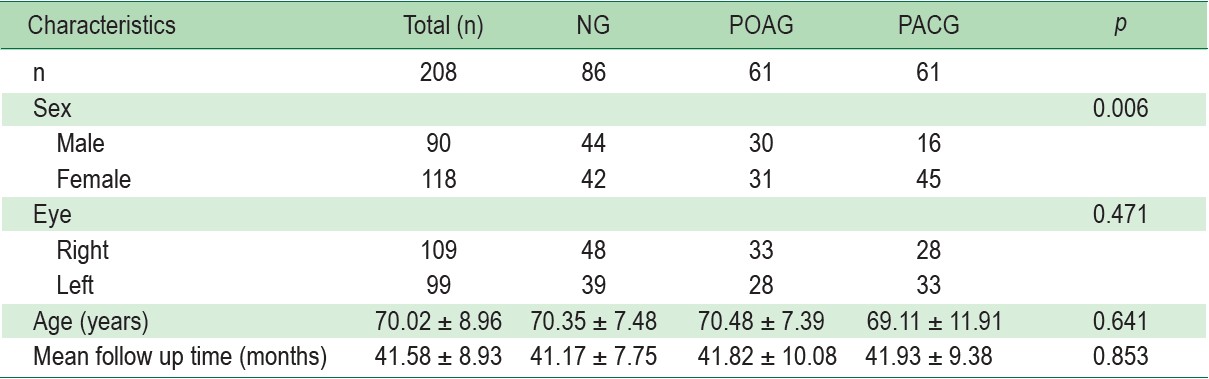
Table 3, 4, and 5 demonstrate the magnitude of IOP decrease in various ranges of preoperative IOP. All the eyes were divided into 3 groups base on preoperative IOP; from 9 to 13 mmHg, 14 to 19 mmHg and 20 to 25 mmHg. The most considerable decrease in mean IOP was found in the group with highest preoperative IOP decrease by 2.06 ± 2.11 mmHg (13.5%) in NG, 4.75 ± 2.82 mmHg (22.2%) in POAG and 6.6 ± 2.59 mmHg (29.9%) in PACG. In contrast, we found that in the group with the lowest preoperative IOP, the postoperative IOP remained unchanged or increased.
The number of antiglaucoma medications in the POAG and PACG group had changed significantly by the final visit. In POAG group, medication decreases from 1.23 ± 0.46 to 1.08 ± 0.53 (p < 0.002) and in PACG group, medication decreases from 1.39 ± 0.67 to 1.08 ± 0.64 (p < 0.001).
The final post-operative mean LogMAR visual acuity improved in all patients (p < 0.001). At a mean 40 months postoperatively, the mean LogMAR visual acuity had improved by -0.43 ± 0.26, -0.37 ± 0.21 and -0.41 ± 0.23. There was no blindness due to surgery.
Table 2 : Intraocular Pressure (IOP) Change and Mean Intraocular Reduction after Phacoemulsification.
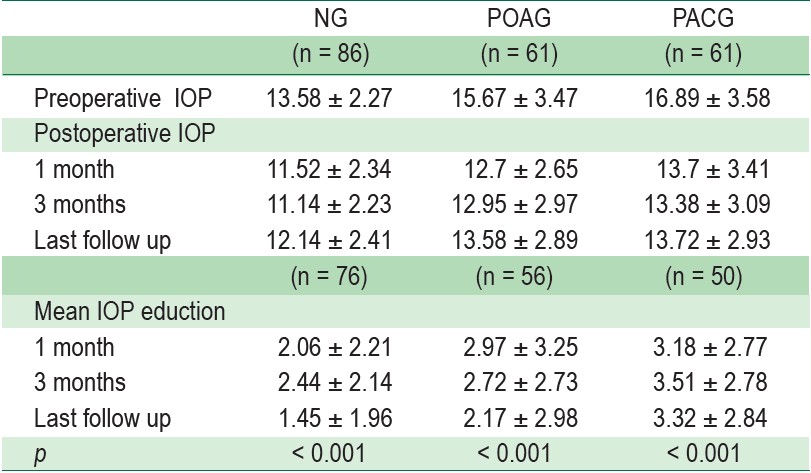
Table 3 : Mean post-operative IOP change based on pre-operative IOP in nonglaucoma group

Table 4 : Mean post-operative IOP change based on pre-operative IOP in POAG group
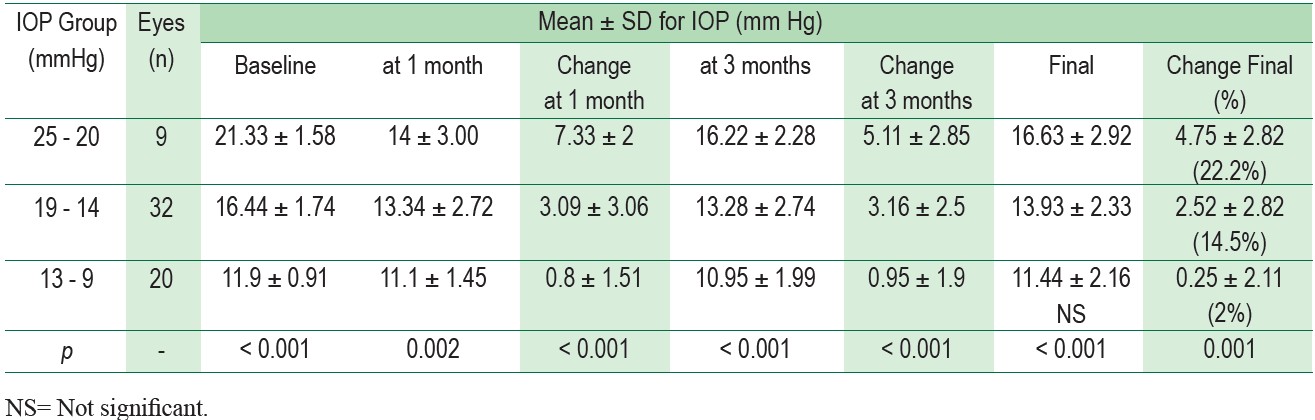
Table 5 : Mean post-operative IOP change based on pre-operative IOP in the PACG group.
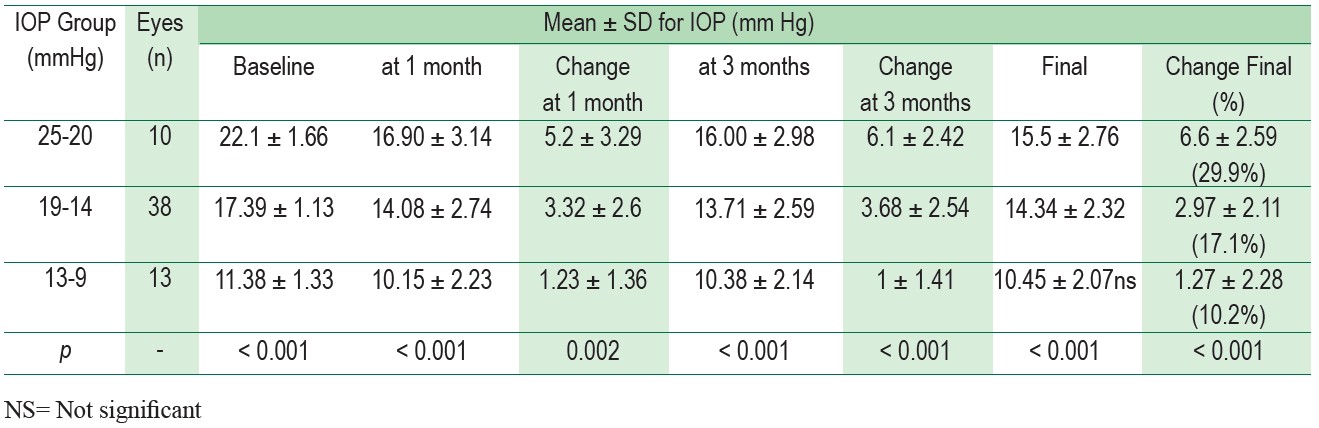
Figure 1: Pre-operative and post-operative mean visual acuity in 3 groups. 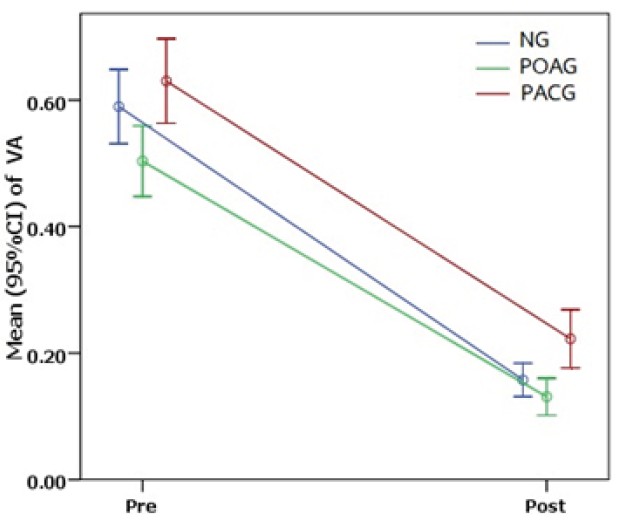
Figure 2: Photographs of a patient with cataract. A: Preoperative. B: One year after phacoemulsification with intraocular lens implantation.
Previous studies have demonstrated the effect of cataract surgery on IOP, and results seems to vary from study to study depending on the type of patient.
Some studies have postulated that cataract surgery is a more effective treatment for PACG patients. However, our research found that phacoemulsification was able to decrease IOP in both non-glaucoma and glaucoma groups. The lowering of IOP in both primary open-angle glaucoma and primary angle-closure glaucoma show no significant difference. Futhermore, pressure is more reduced than in the non-glaucomagroup every time.
Hayashi et al.10 report results in 74 eyes with ACG and 68 eyes with OAG at the 2 years follow up. The decrease in mean IOP from baseline to postoperative was statistically significant in both groups (p < 0.001). The PACG group IOP decreased from 21.4 mmHg to 14.5 mmHg (mean IOP decrease 7.2 mmHg), while the POAG group decreased from 20.7 mmHg to 15.2 mmHg (mean IOP decrease 5.3mmHg). The PACG group mean IOP decreased more than the POAG group (p = 0.02). The number of medications decreased significantly after surgery in both groups (p < 0.0001). This result is similar to our study.
Shingleton et al.7 followed up the POAG patients, glaucoma suspects, and normal patients 3 years after having phacoemulsification. They found that there was a decrease in eye pressure with statistical significance with all 3 groups. The POAG patients showed a decrease in eye pressure at 1.4 mmHg (p = 0.0025), glaucoma suspects a decrease in eye pressure at 1.4 mmHg (p = 0.004), and in normal patients a decrease in eye pressure at 1.7 mmHg (p = 0.0005). There was no change in the amount of antiglaucoma drugs after the surgery. This study postulates that the decrease of IOP is less than that recorded in our study. The different results in this study may be due to enrolled patients with pigment dispersion and pseudoexfoliation in both glaucoma suspects and the glaucoma group. Shingleton et al.18 have shown that there was a decrease of mean IOP in both POAG and PACG patients with statistical significance (p < 0.001) of 1.8 mmHg and 2.1 mmHg at 6 months and 12 months respectively (p = 0.32 at 6 months and p = 0.75 at 12 months). Their result may show less of a drop in IOP than our study, however, the lower preoperative IOP in this study could be responsible for this difference.
Mean IOP reduction in the previous study seems to be moving in the same direction as our study. Nevertheless, there is a difference in the value which may due to the methodology and preoperative IOP status.
In our study, Postoperative mean IOP reduction was 1.45 mmHg in NG, 2.17 mmHg in POAG, and 3.32 mmHg in PACG at the 41 months follow-up. Although this value is statistically significant, it does not appear to be clinically relevant. However, after subgroup analysis, the final mean IOPreduction in the baseline IOP 25 - 20 mmHg for POAG was 4.7 mmHg (22%), and PACG was 6.6 mmHg (29%). This would be a more significant IOP reduction than the overall result. On the other hand, while the baseline IOP is at 13 - 9 mmHg, there was no change from the pressure before surgery which is 0.25 mmHg in POAG (p = 0.643) and 1.27 mmHg in PACG (p = 0.094). Like the study of Poley, et al.19 they have studied the effect of phacoemulsification with intraocular lens (IOL) implantation based on presurgical IOP. They found that the final mean IOP reduction was 6.5 mm Hg (27%) in the 31 to 23 mm Hg and in the 14 to 9 mm Hg group, the mean IOP at the final examination was 0.2 mm Hg higher (0.1% increase). The decrease was most significant in eyes with the highest presurgical IOP and IOP remained unchanged in eyes with the lowest presurgical IOP.
Many theories have been mentioned about the mechanism that cataract surgery causes to reduce intraocular pressure. Kim et al.20 used anterior segment optical coherence tomography in the study of anterior chamber depth and angle parameters to compare angle-closure glaucoma and open-angle glaucoma before and after phacoemulsification. The angle width and ACD in eyes with glaucoma increased significantly after phacoemulsification and IOL implantation. Thus, the result is similar with Siak et al.9 as they found the increase of angle parameter after phacoemulsification in POAG and PACG patients. Another study by Zhao et al.21 used swept-source optical coherence tomography in cataract patients after having cataract surgery as they found that there an increase of Schlemm’s canal area, Schlemm’s canal diameter and trabecular meshwork length resulting in an increase of outflow facility. The recent study by Alaghband et al.22 studied outflow facility with electronic Schiotz tonography and found phacoemulsification enhancement of the facility of outflow. The decrease of postoperative IOP might be caused by both anatomical change of the anterior chamber and the increasing of outflow facility. However, the actual mechanism is still unknown.
From our study, the persistent long-term reduction of IOP after phacoemulsification with intraocular lens implantation has the benefit of decreasing medication in the glaucoma group. Although current antiglaucoma drugs are able to control eye pressure well, they have both local and systemic side effects especially if multiple drugs are used. The failure rate in the trabeculectomy was 46.9%, and tube-shunt was 29.8% at 5 years follow up.23 Furthermore, there were severe complications such as persistent corneal edema and retinal detachment.24 At the very least, phacoemulsification and IOL implantation can reduce subsequent glaucoma surgery. The replacement of the cataractous lens with intraocular lens makes the diagnosis of retina and optic disc easier. It also decreases the opportunity of having high ocular pressure and sudden acute angle-closure from cataracts in the future. However, phacoemulsification is not suitable for every glaucoma patient because many factors need to be considered, such as target IOP, stage of the disease, and patient safety.
The limitation of this study is that it is a retrospective study, based on medical records, with a limitation of available data collection, and there was a selection bias given that a number of patients did not follow up due to relocation and death.
Phacoemulsification can decrease intraocular pressure in normal patients and glaucoma patients without depending on angle status at both one month and three months as well as at the follow up of 41 months. The IOP reduction was greater in the eye with higher preoperative IOP. Later on, after surgery patients are using fewer glaucoma medications and have better vision. Further studies emphasize the need for an extensive, prospective analysis to evaluate the long term effects of IOP-lowering with an assessment of other outcomes (disease progression by visual field loss, quantitative optic nerve analysis or quality of life).