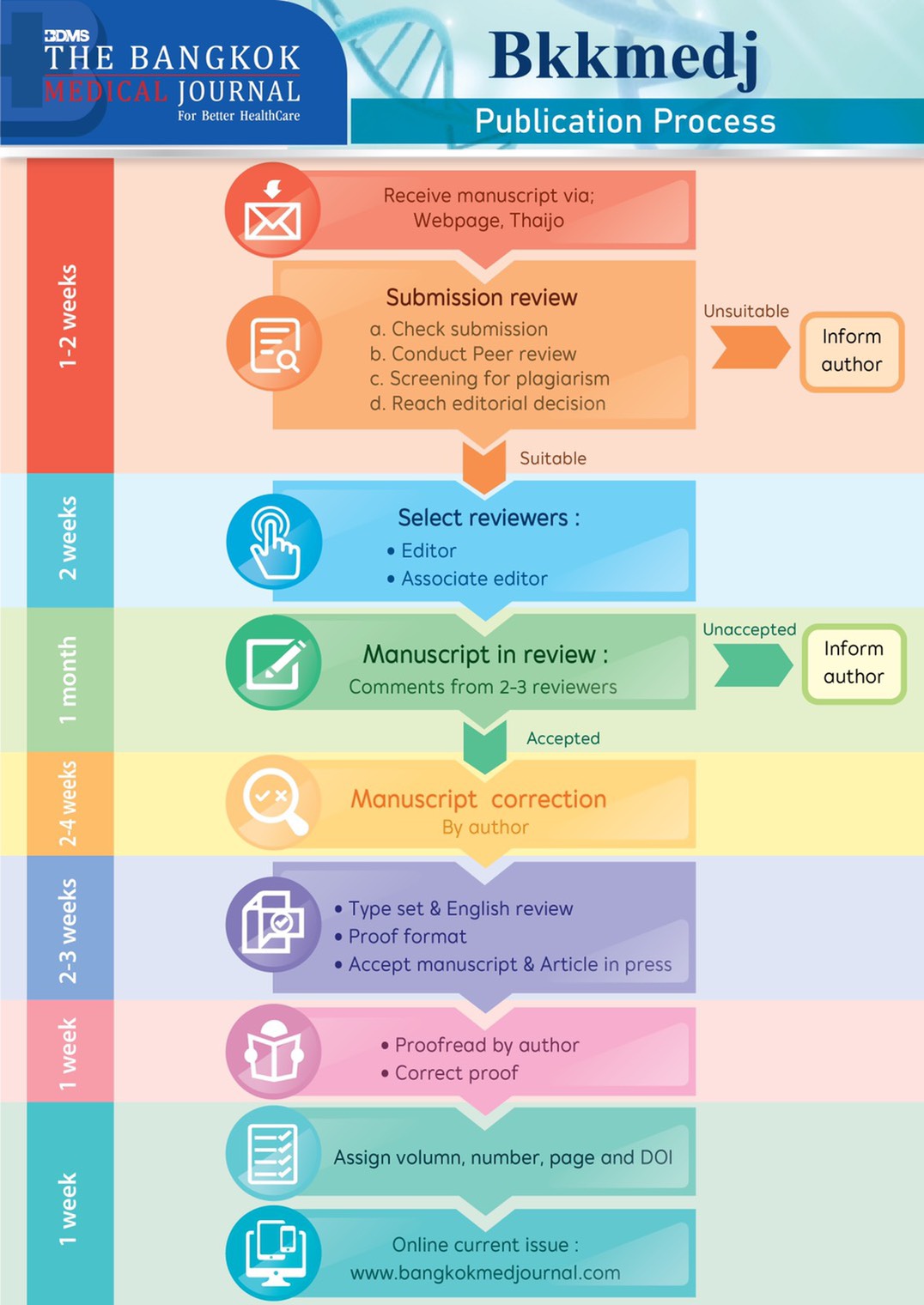
The Editor-in-Chief of The Bangkok Medical Journal invitessubmission of manuscripts in all areas of medical research. As ageneral guide, manuscripts will be considered for publication if theycontribute significantly new findings to the health or medical field.Interested authors are invited to submit their manuscripts throughour Open Journal System (OJS). Manuscripts that are conformed toJournal’s scope and format will go through plagiarism check anddouble blinded peer review process before going for graphic designand final proof.
The Journal is published twice a year, available in February and September and is designed for all physicians, researchers, and healthcare workers regardless of their specialties.
The Bangkok Medical Journal (ISSN 2287-0237 (online) /2228-9674 (print)) is a peer-reviewed journal published by BDMSFoundation for medical education and research. The primary goalis to serve as an educational tool for improved medical practices.The Bangkok Medical Journal accepts original articles dealing withclinical and laboratory medicine, clinical research, basic scienceresearch, and epidemiology. Prospective authors should consult“Instruction for Authors,” at www.bangkokmedjournal.com
Intellectual property is a serious concern of The Bangkok Medical Journal. On submission, all articles are screened using the software ‘iThenticate’. Plagiarism is suspected when the percentage of similarity is higher than 20%. Clear plagiarism (use of large portions of copied text and/or data) results in rejection while minor copying of short phrases leads to a gentle notification to the authors for re-writing. Manuscripts that pass plagiarism check will be moved further as described in the section “Review Process”. If plagiarism is detected later during the review process by any means, the review will be immediately withheld and the Chief Editor will contact the corresponding author and/or all the authors for an explanation. Rejection of submission will occur once the explanation is not satisfied or when the authors’ confession of guilty. After publication, the article will be retracted if plagiarism is proven. The Journal will contact authors’ institutions to notify the retraction and express expectation of relevant behaviors. The action of retraction will be officially announced as early as possible through The Bangkok Medical Journal online and hard copy publications.
The Bangkok Medical Journal follows the guidelines of theInternational Committee of Medical Journal Editors (ICMJE). Theguidelines may be viewed at http://www.icmje.org/icmje-recommendations.pdf
To maintain academic integrity, The Bangkok Medical Journalallows only evidenced-based advertisement that contributes tomedical advancement. Advertisers are responsible for obtainingapproval for advertisement according to legislations of Royal ThaiGovernment or relevant countries where the Journal is distributedto. The Bangkok Medical Journal does not accept advertisementsthat violate medical community standards, appeal any kind ofdiscrimination or related to health hazards including alcohol,tobacco, firearms and any unsafe practices. Decision to accept orreject an advertisement is made by the Editor-in-Chief with guidanceof the Editorial team and selected board members.
The following types of material may be considered forpublication:
1.Original Article: Original Articles are scientific reports of the results of original clinical research.
2. Case Report: Brief Reports usually describe one to three patientsor a single family with an uncommon history or diagnosis or atreatment outcome.
3. Review Articles: Review articles are usually solicited by theeditors, but we will consider unsolicited material. All reviewarticles undergo the same peer-review and editorial process asoriginal research reports. They should be written for the generalphysicians not specialists. Consequently, they may includematerial that might be considered too introductory for specialistsin the field being covered.
4. Medical Images: Images in clinical medicine are classic imagesof common or unusual or rare medical conditions. This featureis intended to capture the sense of visual discovery and variety thatphysicians experience. Medical Images are not intended as avehicle for case reports. Original, high-quality images areconsidered for publication (subject to editing and abridgment)provided they do not contain material that has been submittedor published elsewhere.
5. Letter to Editor: Letters to Editor provide a forum for readersto comment about articles recently published in the Journal, andthey are a place to publish concise articles, such as reports ofnovel cases.
6. Book Review: Book review allows readers to learn overall contentof selected books along with some scientific opinions of the authorswho review those books. This helps readers to appreciate overallcontent before making a decision to go for thorough reading.
7. Special Features: Articles that do not fall into any abovecategories but are considered highly potential to contribute toadvancements in medical or health communities will beaccepted as Special Features.
Article Types
The Bangkok Medical Journal publishes specific type ofarticles. Each type serves a distinct and separate purpose and isjudged by different criteria. Manuscripts should be constructedaccording to specific guidelines for Articles.
To submit each type of manuscript for publication in theJournal, please follow the specific submission instructions at www.bangkokmedjournal.com
Includes the following information:
(1) Cover letter All manuscripts should be submitted with a coverletter, addressed to the Editor-in-Chief, which explains why themanuscript should be published in The Bangkok MedicalJournal. In particular, authors should identify novel findings,innovative approaches, and important insights that would makethe manuscript of particular value to the readers of The BangkokMedical Journal.
(2) Text, table and figure files Every file should be given a labelthat includes the first author’s last name and the nature of the file.
(1) Proofs
Proofs are mailed electronically in a PDF format, andmust be returned within three days of receipt. Late returns of proofswill cause substantial delay in article publication. It is thecorresponding author’s responsibility to see that the proof isaccurately checked and corrected, and to return the proofspromptly to avoid publication delays. Please check the spelling ofco-authors’ names, text, tables, legends, and references carefully.It is the authors’ responsibility to make sure that the informationis accurate. Indicate corrections either using the PDF editor functionor with clear hard-copy indications. The proof corrections stageis not the time for fine-tuning language or making any othersubstantive changes. Confine corrections to errors in printing;authors may be charged for major author-initiated changes.
(2) Public access of accepted/published articles
Public access of accepted/published articles: Prior to acceptance,articles may be shared (printed or electronic copies) withcolleagues. After an article is accepted, authors may shareprinted or electronic copies of the article (accepted and revisedto address peer review) with colleagues, and may use thematerial in personal compilations with other publications of his/her own work, and for educational/research purposes. Articlespublished in The Bangkok Medical Journal are freely accessibleto the public - via website “www.bangkokmedjournal.com
Once an author has prepared his/her manuscript that isconformed to the instructions for authors.
Go to www.bangkokmedjournal.com. First-time authors willneed to register with our Open Journal Submission (OJS) beforesubmitting his/her manuscript on “submit an article”. This will takehim/her to the online submission and peer review portal.
Follow the online instructions for filling out the submissionform and uploading the manuscript.
The author may submit his/her manuscript using filling out thesubmission from and uploading your manuscript.
The Bangkok Medical Journal emphasizes that for researchinvolving human subjects, authors must express their complianceto Good Clinical Practices (GCP) guidelines. Certificate ofApproval (COA) of relevant Institutional Review Board (IRB) orEthics Committee (EC) must be presented along with submittedmanuscripts of studies involving human subjects. In the absence ofthe Certificate of Approval, the authors have to convince theEditorial Board that the study did not violate any ethical concernson human subject research.
Research involving animals must comply with institutional,national or internationally-accepted standards, and whereavailable should obtain prior approval from their InstitutionalAnimal Care and Use Committee (IACUC) or appropriate ethicscommittee. The manuscript must include the name of theapproval ethics committee and the standards that were followed.
The Basel Declaration outlines fundamental principles to adhereto when conducting research in animals and the International Councilfor Laboratory Animal Science (ICLAS) has published ethicalguidelines. Also the Weatherall report on the use of non-humanprimates in research provides recommendations on non-humanprimate studies.
The Bangkok Medical Journal adopts CC BY-NC-ND 4.0 license for its content.
You are free to share copy and redistribute the material in any medium or format
Under the following terms:
Ethical publication ethics stems from ethical research and is augmented with ethical review. The Bangkok Medical Journal thus accepts manuscripts of human research that follow the World Medical Association (WMA) Declaration of Helsinki (2008) and comply with 2016 International ethical guidelines for health-related research involving humans for clinical research provided by The Council for International Organizations of Medical Sciences (CIOMS) and, when relevant, the WMA Declaration of Taipei. As a result, the authors are responsible to demonstrate sufficient compliance to ethical principles by showing a document of approval from the relevant Ethics Committee (EC) or Institutional Review Board (IRB). At the same time, all parties involved in the publication process, namely reviewers, journal editors and journal staff are obligated to follow the Principles of Transparency and Best Practice in Scholarly Publishing guided by the Committee of Publication Ethics (COPE).
Ethics of authors
The authors must present original works, and if they have to use the work or words or others, those work or words are appropriately cited or quoted. Authors must acknowledge previous presented results by citing those works in the manuscripts and listing them in the references. The Bangkok Medical Journal reserves the rights to use plagiarism detection software to assess degree of possible plagiarism at the screening and reviewing processes. Data and results must be accurately presented without fraud, fabrication, falsification or inappropriate manipulation. Discussions and conclusions should be evidence based. If personal opinions are presented, they must accompany with justification and well labeled as personal opinions.
Authors must ensure that statements made in the manuscripts, including changes made by the editors or English language manager, once accepted by the author are the responsibility of the authors.
Once the author is aware of a significant error or inaccuracy in his/her published work, it is the author’s responsibility to immediately notify the Journal editor and cooperate with the Journal editors to retract or correct the paper.
Authors are responsible to disclose financial support and conflict of interest. All existing or potential conflict of interest such as employment, consultancy, stock ownership, an honorarium, paid testimony and intellectual property registration should be disclosed at submission stage. Technical, operational or management supports should be expressed in the acknowledgement.
Ethics of Editors
All manuscripts are treated as confidential documents. Therefore, editors, editorial board members, and Journal staff must not disclose any information obtained from a submitted manuscript to anyone other than the corresponding author, reviewers, potential reviewers, editorial advisers and the publisher. To ensure confidentiality and minimize biases, The Bangkok Medical Journal employs double-blinded peer review process where neither the authors nor the reviewers know each other’s identity throughout the review process.
Unpublished materials in all submitted manuscripts must not be used in any of editor’s own research without a specific written consent of the author(s).
Decisions on publishing submitted manuscripts are made objectively according to recommendations made by reviewers and professional opinions of editors in particular the importance and potential contribution of the article to scientific communities. Editors must withdraw themselves from the review process if they have a conflict of interest or personal stake in the publication of submitted manuscripts.
Ethics of Reviewers
Ideas obtained through peer review process must be kept confidential by the reviewers and must not be used for personal benefit in any forms. In addition, reviewers must not discuss aspects of the work under review with other researchers until the article is published. Unpublished materials in all submitted manuscripts must not be quoted or referred or used in any of reviewers’ own research without a specific written consent of the author(s), which are requested through the Chief Editor or Co-Editor.
Once the reviewer realizes, either before or after receiving a manuscript for review, that he or she know the authors or has been involved in the research described in the manuscript, or has any condition that might bias his or her judgment or recommendation, the reviewer should inform the editors and decline to be reviewer of that manuscript. Potential conflicts of interest, including competition, collaboration, or other relationships or connections with any of the authors, affiliations, or institutions connected to the manuscript must be disclosed to the editors. It is, therefore, the responsibility of the Journal to seek appropriate actions, including seeking more independent reviewers.
Manuscripts must be reviewed objectively in the context of the reviewer's expertise in the field considering potential contribution of the article to the existing research in its field, the quality of research, articulation of the argument, and the strength of the evidence provided. Personal opinions without backing evidence should not be used as criteria for reviewer’s decisions.
Reviewers should, at the best of their capacity, advise relevant work that has been presented but not cited by the authors. A reviewer should alert the editors upon any substantial similarity or overlap between content of the manuscript under review and any other work published or presented elsewhere
Conflicts of interest
Publication Decision
The Bangkok Medical Journal discourages parallel submission. Submitted manuscript that is under consideration for publication by another journal will be immediately rejected.
The editorial team of Bangkok Medical Journal (BKKMEDJ) is responsible for deciding which of the manuscripts submitted to the journal should be published. The editor may be guided by the policies of the journal's editorial board and compliance to legislation requirements as well as social norms and ethics. For example, research that could result in promotion of violence, trafficking, illicit drug use, weapons or negative political consequences might not be accepted for publication. The Chief Editor may consult other editors or reviewers in making this decision.
All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopy, recording, or any information storage and retrieval system, without written permission from the publisher.
Published by: Bangkok Dusit Medical Service, Plc.
Designed by: Ideol digital print co.,ltd.
Printed by: Ideol digital print co.,ltd.
ISSN 2287-0237(online) 2228-9674(print)
